 Posted on
Posted on

Public comment has ended for a proposed rule that would establish additional traceability recordkeeping requires under the Food Safety Modernization Act (FSMA). If approved, this new rule would help the FDA quickly identify who received adulterated food in order to further prevent foodborne illness outbreaks.
The proposed rule would apply to anyone who manufactures, processes, packs, or holds foods on the Food Traceability List (FTL). Those subject to the rule would need to establish and maintain records containing Key Data Elements (KDEs) associated with different Critical Tracking Events (CTEs).
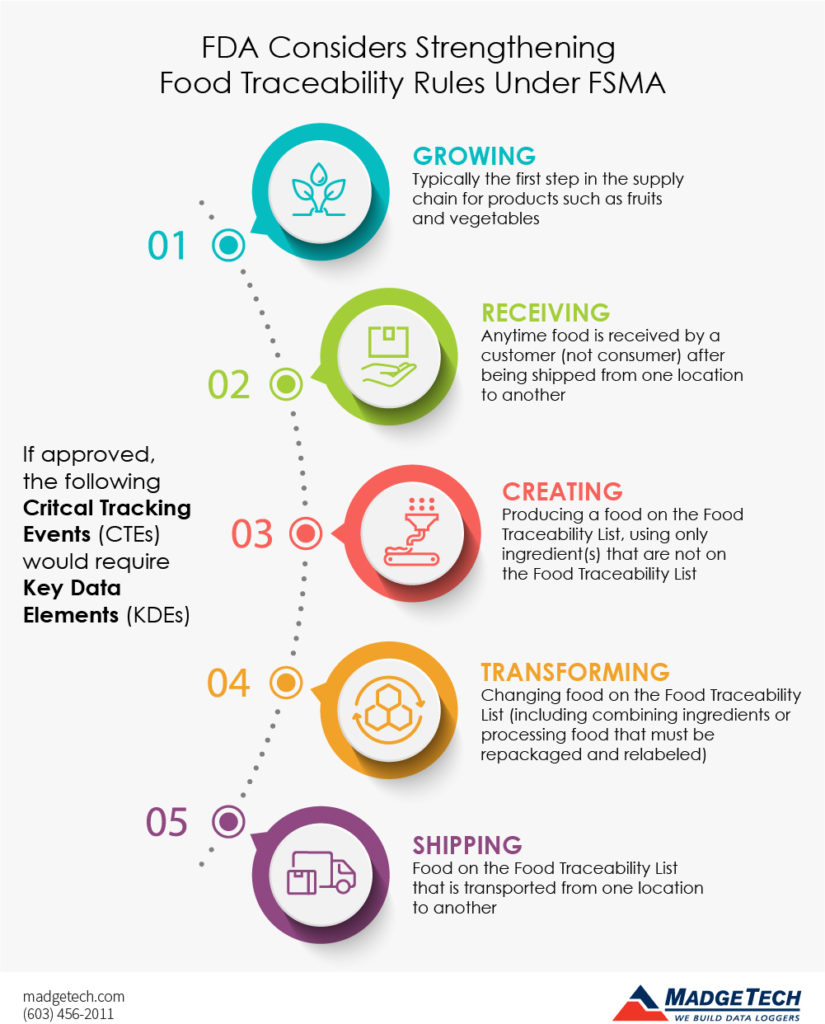
The records would require KDEs for the following five identified CTEs:
1. Growing
Typically the first step in the supply chain, growers would have to link the traceability lot code of the food to the growing area coordinates. Sprout growers would have to provide additional KDEs.
2. Receiving
Anytime food is received by a customer (not consumer) after being shipped from one location to another. The receiver must link the traceability lot code to the general KDEs. The first receiver who purchases the food must maintain additional KDEs.
3. Creating
The manufacturing or processing of food on the Food Traceability List, using only ingredient(s) that are not on the Food Traceability List. Records must link the traceability lot code to date and location details where the food was created, as well as all related reference records.
4. Transforming
Not to be confused with creating, transforming involves changing food on the Food Traceability List. This includes combining ingredients or processing food that must be repackaged and relabeled. Records must link the new traceability lot code of the food produced through transformation.
5. Shipping
Food on the Food Traceability List that is transported from one location to another must link the traceability lot code(s) to shipping KDEs. In addition to keeping these records, the shipper needs to send these records to the recipient.
It’s important to note that there are exemptions for certain food types and persons under the proposed rule. If the final rule is approved, it would become effective 60 days after it is published in the Federal Register. All persons subject to the rule would be expected to be in compliance 2 years after the effective date of the final regulation.
To ensure the quality and safety of food throughout all five CTEs, continuous temperature monitoring through the use of data loggers is required. Designed to fit into any HACCP plan, MadgeTech specializes in data logging solutions for food processors. By streamlining critical data to the MadgeTech 4 Software, reports are automatically generated and saved for analysis and recordkeeping.
To learn more about the benefits of implementing MadgeTech data loggers into your HACCP plan, call us at (603) 456-2011 or email [email protected].






