 Posted on
Posted on
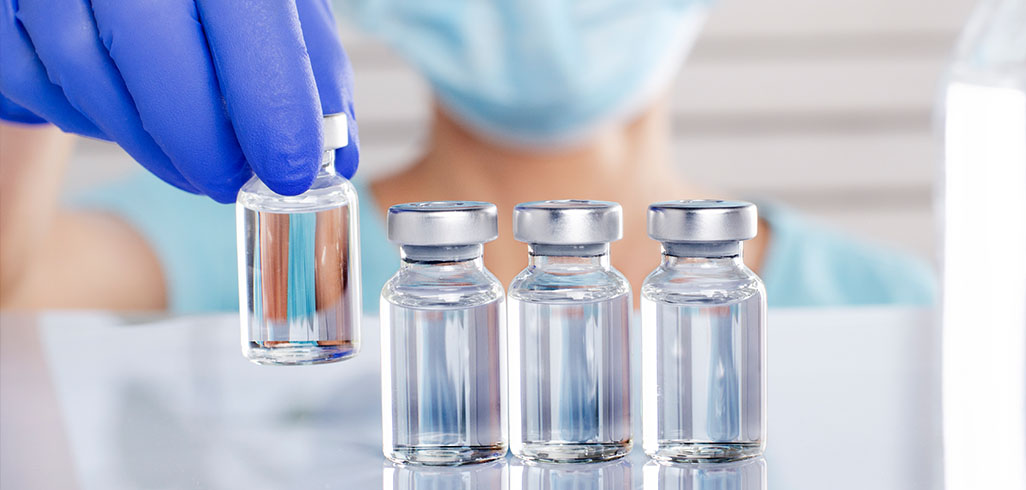
All vaccines are temperature sensitive and require the proper environmental conditions to ensure vaccine stability. Exposure to extreme heat or cold can be catastrophic to millions of vulnerable people, especially children, who would otherwise go unvaccinated.
To ensure the safety and potency of vaccines, the VFC Program, which provides vaccines to children at no cost, has strict requirements in place for its 44,000 providers. In order to participate in government inoculation programs, providers are held to the highest of standards to avoid revaccination, wasted vaccines, and financial loss.
One tool that is crucial to validating vaccine safety and is required for VFC compliance, is a digital data logger (DDL). DDLs provide real-time temperature readings for continuous monitoring during vaccine storage and handling. It’s important to note that not all DDLs are suitable for vaccine temperature monitoring.
Here are the seven features the CDC requires in a DDL being used specifically for vaccines:
- Detachable probe that best reflects vaccine temperatures (e.g., a probe buffered with glycol, glass beads, sand, or Teflon®)
- Alarm for out-of-range temperatures
- Low-battery indicator
- Current, minimum, and maximum temperature display
- Recommended uncertainty of +/-0.5° C (+/-1° F)
- Logging interval (or reading rate) that can be programmed by the user to measure and record temperatures at least every 30 minutes
- ISO 17025 certificate of calibration
DDLs are the most accurate way to measure temperature and are designed to streamline data with ease. The constant stream of data allows users to detect deviations and take corrective actions before it’s too late.
MadgeTech engineers and manufactures data logging systems specifically for vaccine temperature monitoring. The VTMS and VFC2000 are 100% compliant with all CDC and VFC requirements and can withstand ultra-low temperatures necessary for certain vaccines.
To find the right data logging solution for your vaccine management plan, call us at (603) 456-2011 or email [email protected]






