Life Sciences
Showing 1–12 of 48 results
-

HiTemp140
$539.00 – $589.00 Select options -
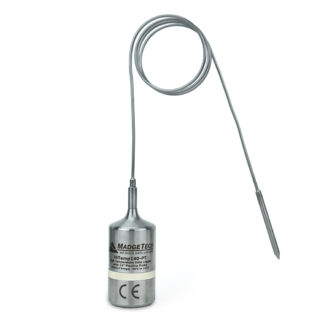
HiTemp140-PT
$539.00 Select options -

RFRHTemp2000A
$349.00 Add to cart -

Titan S8
$999.00 Add to cart -

Titan S8-CAN
$999.00 Select options -

VTMS
$339.00 – $359.00 Select options -

HiTemp140-M12
$599.00 Select options -

PR140
$1,049.00 Select options -

RFTCTemp2000A
$329.00 Select options -

Temp101A
$79.00 Add to cart -

MicroRHTemp
$349.00 Add to cart -

RHTemp101A
$159.00 Add to cart
Showing 1–12 of 48 results
Not what you
were looking for?
were looking for?
or call
(603) 456-2011
(603) 456-2011







